Our Pipeline | Zotatifin & Tomivosertib
Through the joint efforts of our team and our global partners, we are committed to continuously advancing our compounds.
In addition to clinical-stage programs like Zotatifin & Tomivosertib, we are advancing a diversified portfolio of early-stage projects spanning PHB1/eIF4A small molecules and Antibody-Drug Conjugates (ADC) payloads, CDK8 inhibitors, ERK3 modulators, and antiviral research.
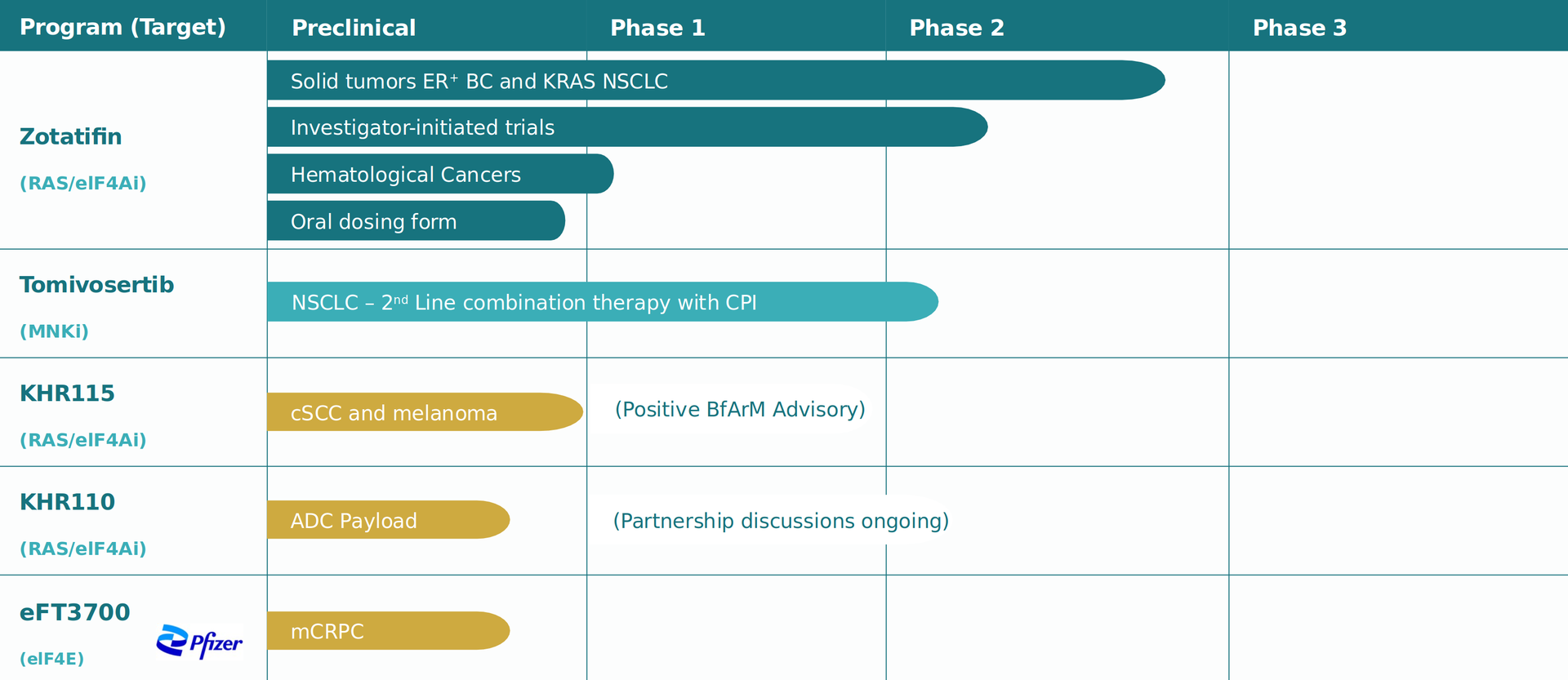
Zotatifin (eFT226) — Lead Asset
Zotatifin (eFT226) is SJP Biotec's lead compound and a first-in-class eIF4A inhibitor derived from flavagline chemistry. It is currently in multiple Phase 2 clinical trials across oncology indications; Zotatifin simultaneously inhibits RAS signalling and translation initiation (eIF4A), blocking two key pathways that tumour cells rely on for growth and survival.
• ER+/HER2− Breast Cancer (triplet combination) — FDA Fast Track Designation
• Diffuse Large B-Cell Lymphoma (DLBCL) — Phase 2, in combination with CAR-T cell therapy
• Mantle Cell Lymphoma — Phase 2
• Pancreatic Cancer (PDAC) — Phase 2
• COVID-19 (SARS-CoV-2) — Phase 2 antiviral application
Tomivosertib - Best-in-Class MNK1/2 Inhibitor
Tomivosertib inhibits Mnk1 & Mnk2 at low nanomolar concentrations with high specificity and is addressing key resistance mechanisms. PK-PD target phosphorylation decreased at all doses tested in man, with good dose-response correlations. It is also orally available and crosses the blood-brain barrier. Potential development options are in oncology, neuropathic pain, Fragile X syndrome, & autism spectrum disorder. The study design was changed in will be continued shortly.
Early-Stage Pipeline
In addition to our clinical programs, SJP Biotec is advancing a diversified portfolio of preclinical and IND-ready drug candidates:
• PHB1/eIF4A Small Molecule Inhibitors — next-generation compounds for solid tumours and haematological cancers
• ADC Payloads (Antibody-Drug Conjugates) — precision-targeted cytotoxic agents for cancer with minimal off-target toxicity
• CDK8 Inhibitors — targeting cyclin-dependent kinase 8 for transcriptional reprogramming in cancer
• ERK3 Modulators — novel modulators of the ERK3 kinase pathway implicated in tumour immune evasion
• Antiviral Research — broad-spectrum antiviral small molecules with applications beyond oncology
• 1 IND-Ready Molecule — preparing for first-in-human clinical trials